Major Banana Diseases Caused by Fungi
I. Black Leaf Streak (Black Sigatoka)
This is the most economically important leaf disease of banana. It destroys banana leaves which results to yield reduction and premature ripening. It is more virulent, has a wider host range, more rapid development and severe defoliation than yellow sigatoka.
Black Leaf Streak (Black Sigatoka)
Causes:
- The fungus Mycosphaerella fijensis
- Influenced by humidity, temperature, leaf wetness, intensity of infection, plant vigor and light intensity.
- Stage 1: Small (less than 1 mm long) whitish or yellow specks on the underside of the leaf, which turn rusty brown.
- Stage 2: Specks grow into narrow (2 to 5 mm long), reddish or dark brown streaks on the underside of the leaf, with corresponding yellow streaks on the upperside.
- Stage 3: Streaks become longer (20 to 30 mm long) and may coalesce to cause leaf necrosis.
- Stage 4: Individual streaks broaden to form brown spots on the underside of the leaf, with corresponding black spots on the upperside.
- Stage 5: Elliptical black spots, usually surrounded by a yellow halo, on both sides of the leaf.
- Stage 6: Centre of the spot dries and fades to clear gray, surrounded by a black border and a water soaked or yellow halo.
An infected leaf
- The leaves turn brown-black, dry quickly and die within 1 to 4 weeks.
- Symptoms usually first seen on leaves 3 and 4, later also on younger leaves.
- Few or no viable leaves at harvest.
- Reduction in quality and quantity of fruit.
1. Cultural Control for Black Sigatoka.
Reduce inoculum levels on farms by:
- Removing leaves with extensive necrosis.
- Removing or cutting the small areas of necrotic leaf tissue as soon as they appear.
- Maintaining an efficient drainage system; and
- Maintaining adequate balance between distribution and number of plants, to avoid overlapping of foliage.
Very susceptible cultivars include:
- Cavendish, Gros Michel (AAA)
- All known plantain cultivars (AAB)
- ABB cooking banana cultivars (Bluggoe, Pelipita, Saba)
- Mysore AAB, T8 (bred hybrid, AAAA), Pisan Lilin (AA), FHIA-03 (AABB), FHIA-18, FHIA-21 (AAAB),FHIA-25
This disease can be controlled by two types of fungicides:
- 2.1 Protectant fungicides These remain on the leaf surface and do not penetrate the underlying tissues. Approve protectant fungicides are dithiocarbamates and chlorothalonil.
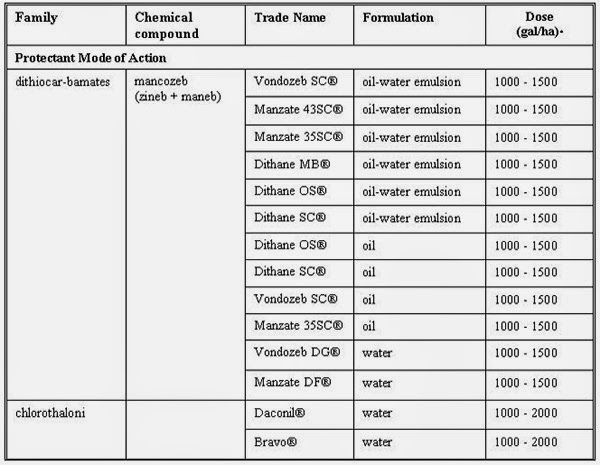
- 2.2 Systemic fungicides These penetrate the leaf and have a specific mode of action, which makes them phytotoxic to banana. They exert a toxic effect on the pathogen after infection has taken place. They belong to the benzimidazole, morpholine, triazole and strobilurin group of fungicides.
- 2.2a Types of Systemic Fungicide Petroleum Oil
- This has been shown to have an effect on disease development. Oil application rates range from 5 to 15 liters per hectare, and it can be applied straight or in an oil-water emulsion. All systemic fungicides are applied in oil. The oil helps the fungicides penetrate the leaves.
- Preventing Build-up of Resistance
- Limit the use of fungicides.
- Base fungicide applications on a disease-forecasting system and/or on crop growth and weather conditions.
- Limit the use of systemic fungicides.
- Always apply systemic fungicides in mixtures with protectant fungicides.
- Alternate between systemic and protectant fungicides.
- Use a rate of oil close to 10 L/ha.
- Do not use reduced rates of systemic fungicides.
- Maintain all cultural practices that will reduce the amount of inoculum.
II. Sigatoka (Yellow Sigatoka)
This used to be the most important foliar disease of banana and caused widespread disruption to the export trade until the discovery and spread of Black Leaf Streak (BLS). Sigatoka causes the premature death of large areas of leaf tissue leading to yield loss and premature ripening.
Cause
- The fungus Mycosphaerella musicola, ascomycete
- Influenced by environmental conditions, host resistance and intensity of infection
- Has a lower temperature optimum than BLS.
- Light green, narrow specks (1 mm long) on the upper surface of the leaf.
- Develops into a streak (several mm long and less than 1 mm wide) running parallel to veins.
- Elongates and expands laterally to become elliptical, turns rusty red.
- Water-soaked halo forms around the lesion and turns brown.
- Centre appears sunken and turns grey, while halo becomes darker brown.
1. Cultural Control for Yellow Sigatoka
Reduce inoculum levels on farms by:
- Removal and destruction of badly spotted leaves (trash).
- Burial or piling of heavily diseased leaves.
- Correct design of blocks to ensure rapid removal of water and good air flow.
- Avoid planting bananas in wet areas or near areas of permanent water.
- Under-tree or or drip irrigation is recommended instead of overhead irrigation.
Very susceptible varieties:
- Cavendish, Gros Michel, Lakatan, Pisang Susu (AAA)
- Sucrier, Inarnibal (AA)
- Plantain (AAB) at elevations
- Yangambi Km 5(AAA)
- Pisang Lilin, Pisang Tongkat, Paka (AA)
- Ney Poovan (AB)
- Plantain (AAB) at sea level
- Bluggoe, Pisang Awak, Kluai Teparot (ABB)
- Application of protectant fungicides before infection.
- Application of systemic fungicides to stop or arrest the fungus after it has infected the leaf (up to the speck or early streak stage, not later).
- Petroleum oil should be included in all sprays since it can arrest infection up to about 2 weeks after it has entered the leaf.
- The frequency of chemical application must be based on:
- disease development, monitored by competent consultants
- weather conditions
- a time-based regular schedule such as:
- Ten to 14 day intervals during wet season;
- Extended to 3 to 4 weeks during dry period; and
- Reduced to 2 to 3 weeks before the next wet season.
- Chemical applications must be aimed at the younger leaves since these are the most prone to new infections.
- Combination of cultural and chemical methods.
- Limited use of systemic fungicides (use when conditions are conducive to infection, not when symptoms are obvious).
- No application of systemic fungicides to heavily diseased plants (de-leaf diseased leaves before applying the systemic fungicide).
- Observation of systemic-free periods.
- Monitoring of the sensitivity of the leaf spot population.
- Limited use of a fungicide from the group to which a particular population of leaf spot is resistant until that particular population has been controlled.
III. Fusarium Wilt (Panama Wilt)
This is regarded as one of the most destructive plant diseases in recorded history. It colonizes the xylem and causes a lethal vascular wilt.
Fusarium wilt of banana from ProMusa Video Bank on Vimeo.
Cause:
The fungus Fusarium oxysporum f.sp. cubense. There are four races of this fungus that attack specific varieties:
- Race 1: attacks Gros Michel (AAA), Maqueno (AAB), Silk (AAB), Pome (AAB), Pisang Awak (ABB), I.C.2 (AAAA)
- Race 2: attacks ABB cooking bananas (e.g. Bluggoe), some bred AAAA tetraploids
- Race 3: attacks Heliconia (before)
- Race 4: attacks same clones as race 1 and 2 + Cavendish (AAA), plantain (AAB), Pisang Mas (AA)
- Subtropical Race 4: plants are usually attacked only when stressed
- Tropical Race 4: can attack unstressed plants
- Infection via root tips.
- In susceptible varieties, the fungus moves in xylem vessels via the plant’s transpirational flux.
- In resistant varieties, the mechanical host defenses to stop systemic colonization of the xylem.
- Dissemination through: infected rhizomes, soil, running water, farm implements and machinery.
- Long-term survival in soil.
- Parasite of non-host weed species.
Internal
- reddish to dark brown discoloration of vascular system
- first in feeder roots and the rhizome and later also in the pseudostem
- yellowing of oldest leaves
- longitudinal splitting of lower portion of the outer leaf sheaths pseudostem splitting
- wilting
- collapse of leaves at petiole base
- gradually younger
- leaves collapse, until the entire canopy consists of dying or dead leaves.
- yellowing starts from older leaves
- symptoms do not develop on plants and suckers less than 4 months old
- no brown discoloration in fruit
There is no cure for Fusarium Wilt. Prevention and immediate eradication once detected is your only option.
Prevention
- Disinfection of farm tools and machinery.
- Use of pathogen-free planting material (tissue culture) in uninfested soil.
- Immediate eradication of diseased plants (6-m radius) and removal of diseased plant parts from the farm.
- Rice hull burning.
- Quarantine measures
- Good drainage system
- Annual cropping
- Natural sources of resistance in wild species and cultivars.
- Synthetic diploids developed by breeding programs (using resistance in Pisang Jari Buaya, Pisang Lilin and Musa acuminata ssp. burmannicoides (Calcutta 4): FHIA-01, FHIA-03 à R r1, r4; FHIA-17 à R r1; FHIA-23 à T
- Somaclonal variant selection (Taiwan): GCTCV 119, GCTCV 215 à R r1

.jpg)







0 Comments